Retinoids (Tretinoin, Adapalene, Tazarotene, Trifarotene) Can Irritate the Skin, Especially in the First Few Weeks, Which Might Make Sunburns Worse

The Essential Info
Retinoids are somewhat effective topical prescription acne treatments that work by unclogging pores and reducing inflammation. The FDA has approved these 4 retinoids:
- Tretinoin
- Tazarotene
- Adapalene [0.1% strength is now available over-the-counter as well]
- Trifarotene – the newest topical retinoid on the market with limited data
You may have heard that retinoids make your skin more sensitive to the sun, and all 4 of these retinoids come with warning sections on their package inserts that emphasize the importance of using sun protection while using them.
However, based on available published scientific evidence, it is unknown whether retinoids make the skin more prone to sunburn. Interestingly, we have one study on adapalene that found that adapalene does not make the skin more prone to sunburn, and one clinical trial whose preliminary results suggest that trifarotene might make the skin more prone to sunburn. But what about tretinoin and tazarotene? We will have to wait for a final answer until this research is published.
In any case, retinoids may cause skin irritation, especially in the first few weeks of use, and it is reasonable to assume that this might make sunburn symptoms worse. Therefore, even with the lack of data, it is always prudent to abide by the package inserts and avoid excessive sun exposure, use sunscreen, or practice sun protection habits like wearing hats and clothing to protect the skin when using retinoids.

The Science
- Recommendations for Retinoids and Sun Exposure
- Do Retinoids Make the Skin More Prone to Sunburn?
- Retinoids Causing Skin Irritation = Sun Sensitivity?
- Summary
Recommendations for Retinoids and Sun Exposure
Retinoids are a class of topical prescription medications that are proven to be somewhat effective in the treatment of acne. They work to reduce acne by clearing clogged pores and reducing inflammation. The 4 approved retinoids used for the treatment of acne are:
- Tretinoin
- Tazarotene
- Adapalene [0.1% strength is now available over-the-counter]
- Trifarotene
All of these retinoids have caution and warning sections regarding sun exposure printed on their package inserts. These warnings state that patients using these retinoids should avoid exposure to the sun, or to use sunscreen and protective clothing if sun exposure cannot be avoided.1-5 The package insert warnings are listed below.

Hard evidence on whether prescription retinoids increase sun sensitivity is scarce, but it is safer to assume that these drugs somehow make the skin more sensitive to the sun since they all come pre-packaged with warnings regarding sun exposure.
There are 2 ways retinoids could potentially make the skin more sensitive to the sun:
- They could make the skin more prone to getting a sunburn
- They could irritate the skin, which might worsen the effects of a sunburn
It’s important not to get frequent or severe sunburns because they can make a form of skin cancer called melanoma more likely. Getting a sunburn does not guarantee that skin cancer will develop, but more frequent sunburns increase the risk of getting skin cancer.6
Let’s explore how retinoids might make the skin more sensitive to the sun.
Do Retinoids Make the Skin More Prone to Sunburn?
There are 2 types of sunburns:
- Normal sunburn: A normal sunburn is a strong inflammatory response of the skin when the skin is exposed to too many of the sun’s ultraviolet B (UVB) rays. These sunburns appear as red, hot, painful, and potentially swollen areas of the skin that were exposed to the sun. Fair-skinned individuals develop sunburns more easily than darker-skinned individuals. It is possible that retinoid could increase susceptibility to sunburn, potentially through skin irritation.
- Drug-induced photosensitivity: Drug-induced photosensitivity is when the use of a medication makes the skin especially prone to sunburn. Photosensitivity is an abnormal and exaggerated reaction that the skin has to the sun. This means that a photosensitivity-induced sunburn causes the skin to burn more easily than it would normally burn. For example, someone experiencing drug-induced photosensitivity could develop a sunburn even if the skin is exposed to the sun for only a short time, or on a cloudy day.7 Retinoids, with the possible exception of trifarotene, do not appear to cause drug-induced photosensitivity.
Expand to read about retinoids and photosensitivity
Most retinoids do not cause drug-induced photosensitivity, but trifarotene might
Drug-induced photosensitivity can cause two types of skin reactions:
1. Phototoxicity: Phototoxicity is a skin irritation that develops within a few hours after sun exposure. This is the most common photosensitivity reaction.
2. Photoallergy: Photoallergy is an allergic reaction that develops several days after sun exposure7
Three of the 4 main retinoids do not appear to cause phototoxicity or photoallergy, which therefore means that they would not cause drug-induced photosensitivity.
When it comes to trifarotene, the latest arrival on the market, one clinical trial suggests that it might cause drug-induced photosensitivity, but more research is needed to draw firm conclusions.
- Tretinoin: A 2009 study published in Photodermatology, Photoimmunology, and Photomedicine found that tretinoin is neither phototoxic nor photoallergenic9
- Tazarotene: The package insert provided with the medication tazarotene states that tazarotene does not induce phototoxicity or photoallergy3
- Adapalene: The package insert for a 0.1% adapalene gel states that 0.1% adapalene does not induce phototoxicity or photoallergy. In addition to not causing drug-induced photosensitivity, according to this package insert, adapalene does not cause an increased risk of normal sunburn either.10
- Trifarotene: The package insert for a 0.005% trifarotene cream describes a clinical trial suggesting that trifarotene might increase photosensitivity. In the clinical trial, 1220 patients with acne used the trifarotene cream for 12 weeks, while 1220 other patients with acne used a similar cream lacking medicinal ingredients (a placebo). In the trifarotene treatment group, 2.6% developed a sunburn, compared to 0.5% in the placebo group. In fact, sunburn was one of the most common side effects of trifarotene.5
Side note: The Food and Drug Administration (FDA) has reported that isotretinoin (Accutane®), which is a retinoid that is taken orally, may lead to photosensitivity. So, keep in mind that although tretinoin, tazarotene, and adapalene do not cause drug-induced photosensitivity, isotretinoin may cause the skin to be more sensitive to the sun.8
Retinoids Cause Skin Irritation – Does This Increase Susceptibility to Sunburn?
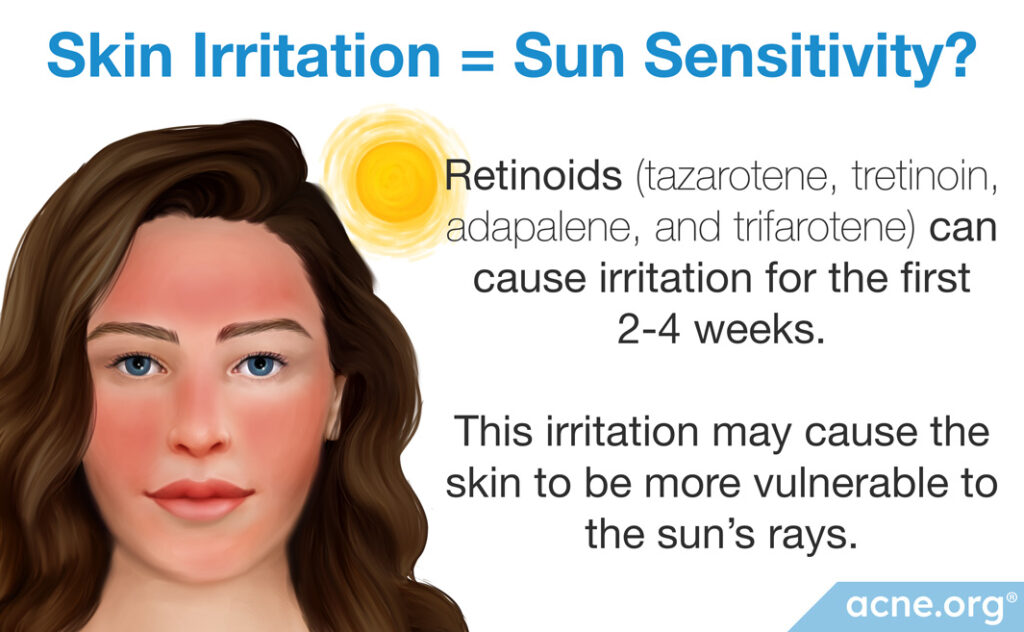
If retinoids do not appear to cause drug-induced photosensitivity, and the package insert of adapalene specifically states that adapalene does not cause an increased risk of normal sunburn, why do the package inserts that accompany topical retinoids insist that people avoid sun exposure or use sunscreens or sun protection during treatment?
One potential explanation may lie in the fact that retinoids are skin irritants, meaning that they irritate the skin and can cause mild-to-moderate skin peeling, redness, or dryness. This irritation is called retinoid dermatitis and is present on the skin for only the first 2 to 4 weeks of retinoid treatment. After that time, the skin becomes accustomed to the retinoid and the irritation subsides.
This means that during the first 2 to 4 weeks of use, retinoids can cause skin irritation, and people with irritated skin may experience increased discomfort if they get a sunburn. This is especially true for acne-prone skin, which is already more sensitive than some other skin types.
One study in the journal Photodermatology, Photoimmunology & Photomedicine puts forth a different theory, and notes that chronic usage of topical retinoids may thin the top layer of skin, which may also make the skin more vulnerable to UV rays.
Expand to read details of study

“Sufficiently chronic use is associated with…thinning of the [uppermost layer of the skin] due to [skin peeling],”9 potentially making the skin more sensitive to the sun’s rays.9
And another study published in the Journal of Cutaneous Medicine and Surgery goes on to note that people with sensitive skin may be more at risk of skin irritation from topical retinoids, and that the higher the concentration of the medication, the more potential for skin irritation.
Expand to read details of study

A 2015 study published in the Journal of Cutaneous Medicine and Surgery compared several studies investigating retinoids and acne in order to determine how irritating tretinoin, tazarotene, and adapalene are to the skin. This study found that tretinoin, tazarotene, and adapalene all caused some skin irritation during the first 2 to 4 weeks of treatment, but that the skin irritation was generally mild. However, the researchers did notice that adapalene gel and cream were the least irritating, and tazarotene gel and tretinoin cream were the most irritating to the skin.
This study also found that the ability to tolerate retinoid treatment with only minimal irritation depended on whether a person normally had sensitive skin. In other words, people who had sensitive skin that was easily irritated when using facial products were more likely to develop skin irritation from retinoid treatments as well. Generally, people with normal skin tolerated retinoids much better than people with sensitive skin. Additionally, products with lower retinoid concentrations were better tolerated than those with higher retinoid concentrations. This means that skin irritation is more common when retinoids are used by people who are prone to skin irritation, used by people with sensitive skin, and used in a product containing a high concentration of retinoids.1,11
When it comes to trifarotene, due to its recent arrival on the market, only 1 study so far has compared the potential of trifarotene to irritate the skin against other retinoids. The study suggests that trifarotene may be more irritating than either adapalene or tazarotene. Tretinoin was not included in the study.
Expand to read details of study

This study was published in the Journal of Dermatological Treatment in 2023. The researchers compared the irritating potential of 0.005% trifarotene cream to 0.3% adapalene gel and 0.045% tazarotene lotion. The study included 40 healthy adults. Each participant applied 3 skin patches to the upper back:
- A patch with trifarotene cream
- A patch with either adapalene or tazarotene, randomly assigned by the researchers
- A patch containing no medicinal ingredients for comparison
Each participant wore the skin patches for 12 days. The researchers removed the patches every 2-3 days, examined the skin for irritation, and applied fresh patches in the same locations.
The scientists found that adapalene and tazarotene were about equally irritating to the skin, while trifarotene seemed to cause the most irritation.12
Additionally, the package insert provides the results of a clinical trial showing that trifarotene is somewhat irritating to the skin. Out of 1220 patients who used trifarotene for 12 weeks, 7.5% experienced skin irritation, compared to only 0.3% of patients who used a similar cream lacking a retinoid. The irritation occurred in the first month of use and decreased with continued use of the medication.5

Summary
Retinoids are a common medication effective in the treatment of acne. Of the 4 available retinoids, trifarotene might cause drug-induced photosensitivity, meaning that it may make the skin more prone to sunburn. The other 3 widely available retinoids (tretinoin, adapalene, and tazarotene) do not appear to cause drug-induced photosensitivity, but they do cause skin irritation, which may aggravate a sunburn.
Out of the 4 topical retinoids, adapalene is the least irritating, while while trifarotene seems to be the most irritating. However, more data on trifarotene is needed before we can draw firm conclusions.
Regardless of whether there is hard data backing up the need to protect one’s skin from the sun while on retinoids, it is always a good idea to abide by retinoid package inserts, which all recommend avoiding sun exposure or using sun protection when using retinoids to treat acne.
References
- Leyden, J., Stein-Gold, L. & Weiss, J. Why topical retinoids are mainstay of therapy for acne. Dermatol Ther 7, 293 – 304 (2017). https://www.ncbi.nlm.nih.gov/pubmed/28585191
- RETIN-A MICRO. Package insert. Valeant Pharmaceuticals North America LLC Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=08ab7e0c-1437-455f-815c-98904d96a289
- TAZORAC® Gel 0.1%. Package insert. Allergan, Inc. Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=75145c21-6ef2-455a-8a67-d48ddd4181a4
- DIFFERIN (adapalene) Gel, 0.3%. Package insert. Galderma laboratories. Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=a0031324-92a6-4c11-90e2-c2818f7278ec
- AKLIEF- trifarotene cream, 0.005%. Package insert. Galderma laboratories. Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=62d910db-85a6-4696-b69b-4bd2f3080cfc
- Dennis, L. K. et al. Sunburns and risk of cutaneous melanoma: does age matter? A comprehensive meta-analysis. Ann Epidemiol 18, 614 – 27 (2008). https://www.ncbi.nlm.nih.gov/pubmed/18652979
- Lim, H. W., Honigsmann, H. & Hawk, J. L. Photodermatology (Informa Healthcare, New York, 2007). https://books.google.com/books?id=AFXOAwAAQBAJ&pg=PA274&lpg=PA274&dq=Lim,+H.+W.,+Honigsmann,+H.+%26+Hawk,+J.+L.+Photodermatology+(Informa+Healthcare,+New+York,+2007).&source=bl&ots=dfBJavDWci&sig=ACfU3U0Uy_C-cti4Kjh_eXIkZOhRzrSrYg&hl=en&sa=X&ved=2ahUKEwjD6rWtjc_lAhXona0KHQ23AkIQ6AEwAHoECAkQAQ#v=onepage&q=Lim%2C H. W.%2C Honigsmann%2C H. %26 Hawk%2C J. L. Photodermatology (Informa Healthcare%2C New York%2C 2007).&f=false
- U.S. Food and Drug Administration. The Sun and Your Medicine. Available from: https://www.google.rs/search?q=The+Sun+and+Your+Medicine&rlz=1C1GCEA_enRS749RS749&oq=The+Sun+and+Your+Medicine&aqs=chrome..69i57j69i64.1059j0j9&sourceid=chrome&ie=UTF-8
- Slade, H. B., Shroot, B., Feldman, S. R., Cargill, D. I. & Stanfield, J. Reappraising the phototoxicity of tretinoin: a report of four controlled clinical trials. Photodermatol Photoimmunol Photomed 25, 146 – 52 (2009). https://www.ncbi.nlm.nih.gov/pubmed/19438994
- Differin® Gel Adapalene 0.1% Topical Gel. Available from: https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/NonprescriptionDrugsAdvisoryCommittee/UCM495797.pdf
- Culp, L., Moradi Tuchayi, S., Alinia, H. & Feldman, S. R. Tolerability of topical retinoids: Are there clinically meaningful differences among topical retinoids? J Cutan Med Surg 19, 530 – 8 (2015). https://www.ncbi.nlm.nih.gov/pubmed/26088502
- Draelos, Z. D. Low irritation potential of tazarotene 0.045% lotion: Head-to-head comparison to adapalene 0.3% gel and trifarotene 0.005% cream in two studies. J Dermatolog Treat 34, 2166346 (2023). https://pubmed.ncbi.nlm.nih.gov/36622889/
 Acne.org Products
Acne.org Products