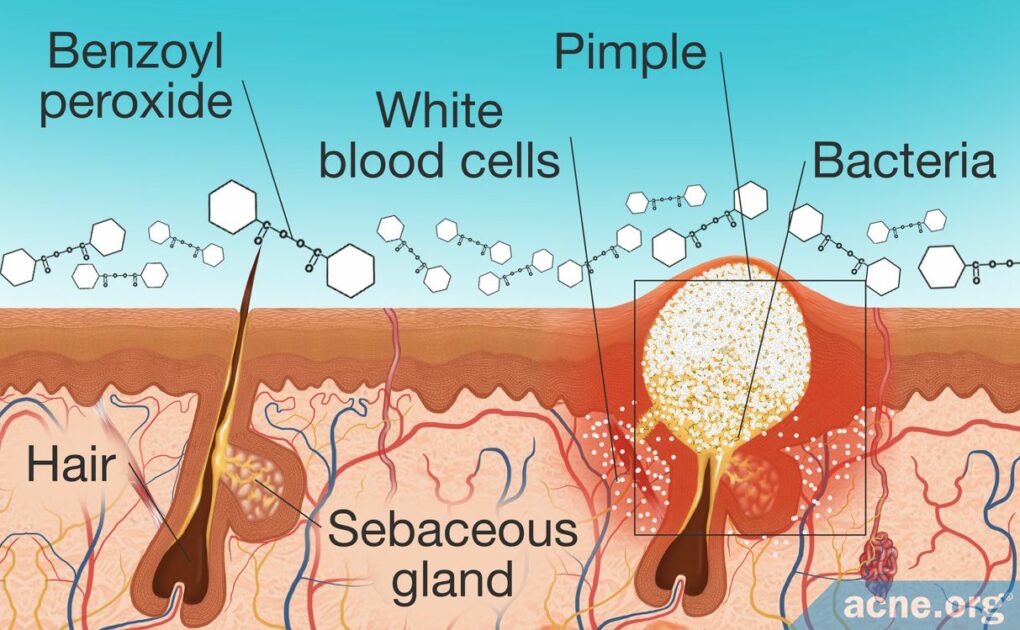
It Kills Bacteria and Unclogs Pores

The Essential Info
Benzoyl peroxide has been used for decades in the treatment of acne and is proven to be effective.
In studies, it reduces acne lesions by 50% on average after 3 months, but when used within a proper regimen, it can completely clear the skin.
When combined with other medications, it also helps them work better.
How does it do this? Benzoyl peroxide is unique because, unlike many medications, it easily penetrates into the skin, where it breaks down into:
- Benzoyl radicals, which kill bacteria and inflammatory cells.
- Benzoic acid, which is thought to promote the opening of clogged pores.
The side effects of benzoyl peroxide are manageable and decrease as treatment continues. Recommendations to minimize these effects are to use a lower dose (2.5% strength), start treatment with a small amount just once a day for the first week, and then begin to slowly ramp up your dosage until you are completely clear. Full instructions here.

The Science
- What Exactly Is Benzoyl Peroxide?
- Action of Benzoyl Peroxide in The Skin
- All the Ways Benzoyl Peroxide Works for Acne
- Side Effects of Benzoyl Peroxide
This article delves into the nitty gritty science behind benzoyl peroxide, its chemical structure, and exactly how it works inside the skin.
Let’s start with its history. Benzoyl peroxide was first used in agriculture and industry. In 1929, physicians discovered that it could be used as a topical treatment for wounds and burns. In 1934, the researchers Lyon and Reynolds found that it was an effective treatment for a case of acne, however it was not until the 1960s that physicians began using it as a regular treatment for acne patients. Now, 60 or so years later, benzoyl peroxide is used as a main treatment for mild-to-moderate acne,1 and when applied correctly can even bring cases of severe acne under control.
What Exactly Is Benzoyl Peroxide?
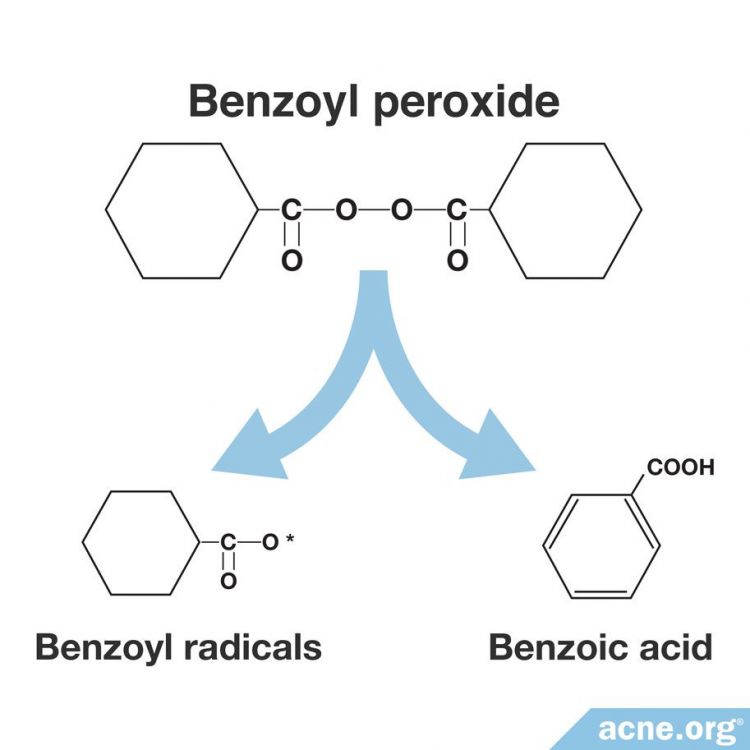
Manufacturers derive benzoyl peroxide from coal tar. After several processes, the final product is in the form of white crystals. It is a compound made out of carbon, oxygen, and hydrogen atoms with most of the carbon atoms linked in two “benzoyl” ring structures.
Once absorbed into the skin, it breaks down into two compounds:
- Benzoyl radicals, which kill bacteria
- Benzoic acid, which helps unclog pores2
Action of Benzoyl Peroxide in The Skin
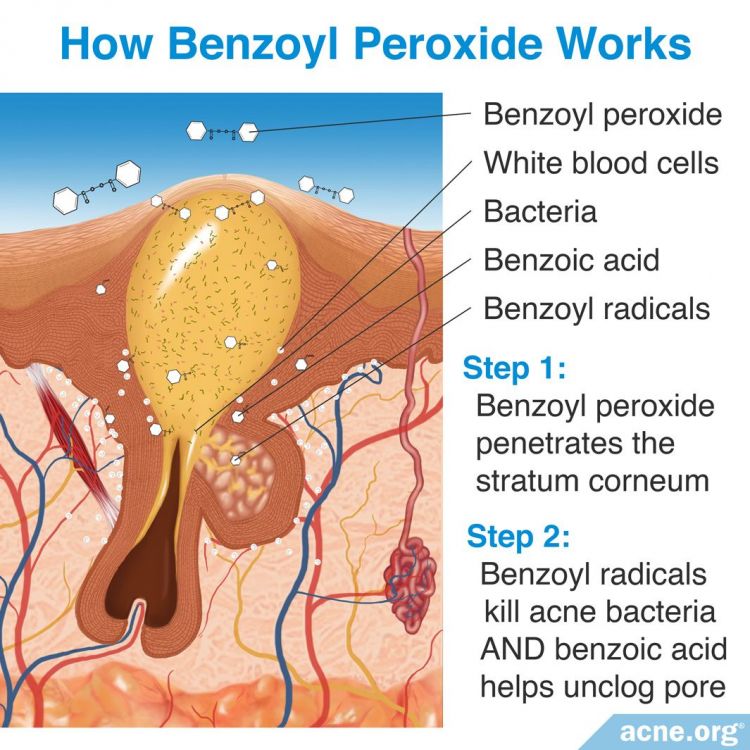
Penetration into the Skin
Benzoyl Peroxide does not dissolve in water but dissolves easily in fats. Thus, when applied to the skin, it easily penetrates the fatty outermost layer of the skin where it then enters the pores where acne is formed.1
Once in the skin, it breaks into its constituent parts: benzoyl radicals and benzoic acid.
Benzoyl radicals get to work fast and kill well over 99% of bacteria. Since acne is in part a bacterial disease, this is thought to be benzoyl peroxide’s main mechanism of action against acne.
Benzoic acid is thought to work similarly to salicylic acid, another well known acne treatment that helps unclog pores. Any extra benzoic acid enters the blood circulation where the kidneys then rapidly metabolize it with no known side effects.3
A Closer Look at Benzoyl Radicals
Benzoyl radicals are a special type of atom with unpaired electrons called free radicals, which create oxygen inside the skin in a process called oxidation.
Acne bacteria can only tolerate short periods of exposure to oxygen, so through oxidation, the free radicals create a hostile environment in which the acne bacteria ultimately die off.
Because benzoyl peroxide is so powerful, its free radicals not only kill bacteria but also potentially cause so-called oxidative damage to the skin. This sounds scary, but fortunately, the skin has defenses called antioxidants to protect itself from oxidative damage. One of these antioxidants is a type of vitamin E called α-tocopherol. Research shows that applying benzoyl peroxide to the skin can deplete the skin’s α-tocopherol by 90%. This is why it’s a good idea to apply an antioxidant-containing moisturizer after using benzoyl peroxide to replenish antioxidant stores in the skin.4,5
Expand to read details of research on BP and antioxidants

The first study was published in the journal Skin Pharmacology and Applied Skin Physiology in 2001. According to the researchers, “It is very likely that barrier α-tocopherol depletion occurs during BP use in acne treatment. Vitamin E depletion and subsequent oxidation of lipids and proteins may account for common side effects such as dry and scaly skin.”4 This is thought to be the reason for the dryness and flakiness that many people experience after benzoyl peroxide application, and is why a moisturizer is usually applied after benzoyl peroxide use.

The second study was published in the Journal of Biochemical and Molecular Toxicology in 2004. This study, which was conducted in a petri dish, confirmed that benzoyl peroxide can cause oxidative damage to the skin. The researchers found that applying vitamin E, which has antioxidant properties, can protect skin cells from this type of damage.5
Benzoyl Peroxide’s Effectiveness
Benzoyl peroxide consistently shows significant effectiveness compared to placebos. The drug shows, on average, a 50% reduction of inflamed lesions and a 46% reduction of non-inflamed lesions.1 This effectiveness rate seems very low to people who use benzoyl peroxide correctly in the real world, however, because when used properly, benzoyl peroxide can completely clear the skin.
All the Ways Benzoyl Peroxide Works for Acne

Kills Acne Bacteria
Acne bacteria, called C. acnes, contribute to the redness, swelling, and pain of acne.
Benzoyl radicals oxidize the skin and disrupt vital biological processes in the bacteria such as DNA and protein synthesis. Benzoyl radicals kill most bacteria that live on human skin quickly and reliably by destroying the cell walls of skin bacteria.2,3,6
Here are some experimental results:
- Twice-daily application of benzoyl peroxide reduces acne bacteria (C. acnes) by 95%.
- 5% benzoyl peroxide significantly reduces the number of C. acnes in only 48 hours.
- Benzoyl peroxide kills the same amount of C. acnes in 3 days that it takes clindamycin (an antibiotic applied to the skin) 14 days to kill.1
Opens Clogged Pores
Keratin is a protein that accumulates within pores, causing the pore to clog.
Benzoyl peroxide breaks up keratin and opens clogged pores.
Several experiments demonstrate that benzoyl peroxide reduces the size of clogged pores by 50%.1 Benzoyl peroxide breaks up keratin better than tretinoin and salicylic acid, two other common acne treatments.3
Promotes Flow of Skin Oil
After application of benzoyl peroxide, researchers notice more oil on the surface of patients’ skin. Benzoyl peroxide does not increase the amount of skin oil that the skin produces, but instead allows the skin oil to escape faster from the pores.
This helps with acne because the flow of skin oil can bring with it the skin cells that would normally stay put in the pore and become a clog. In a sense, benzoyl peroxide irrigates pores.
A 2007 study in the Journal of the European Academy of Dermatology and Venereology give us more detail regarding increased skin oil flow after benzoyl peroxide application:
Expand to read details of study

The researchers speculated that, while the skin continues to produce normal amounts of sebum, benzoyl peroxide allows this sebum to escape to the skin surface faster by unclogging pores.7
Reduces Inflammation
Acne is a chronic inflammatory disease.
A few studies demonstrate the anti-inflammatory properties of benzoyl peroxide. The studies use cell cultures in the lab, however, and not real patients, so it is not certain that the results will be the same for real patients.
These studies show that benzoyl peroxide kills inflammatory immune cells, which prevents these cells from releasing inflammatory molecules in acne lesions.1,3
Combines with Other Treatments for an Enhanced Effect
Benzoyl peroxide combines with other acne treatments to produce an effect greater than the sum of the individual effects. This increase is called a synergistic effect.
The combination of benzoyl peroxide with the antibiotics erythromycin and clindamycin, for instance, increases the antibiotics‘ effectiveness because benzoyl peroxide also kills bacteria. The action of benzoyl peroxide is in turn enhanced by the antibiotics because antibiotics help promote the formation of benzoyl radicals, boosting the bacteria-killing ability of benzoyl peroxide.
However, this is not to say that benzoyl peroxide should be combined with antibiotics. Antibiotics come with serious shortcomings. The first shortcoming is that bacteria can develop resistance to antibiotics. Also, antibiotics should be used for no longer than 3 months, so they are a short-term help at best.
Still, if you are using topical antibiotics, doctors very often add benzoyl peroxide to the treatment regimen, because benzoyl peroxide kills bacteria in a way that does not allow the bacteria to develop resistance.2
The combination of benzoyl peroxide with the popular acne medications in the retinoid family (tretinoin, adapalene, and tazarotene) also creates synergistic effects. The mechanism behind this is less studied. Each medication has its own anti-inflammatory and pore-opening actions and these actions probably reinforce each other.9
Side Effects of Benzoyl Peroxide
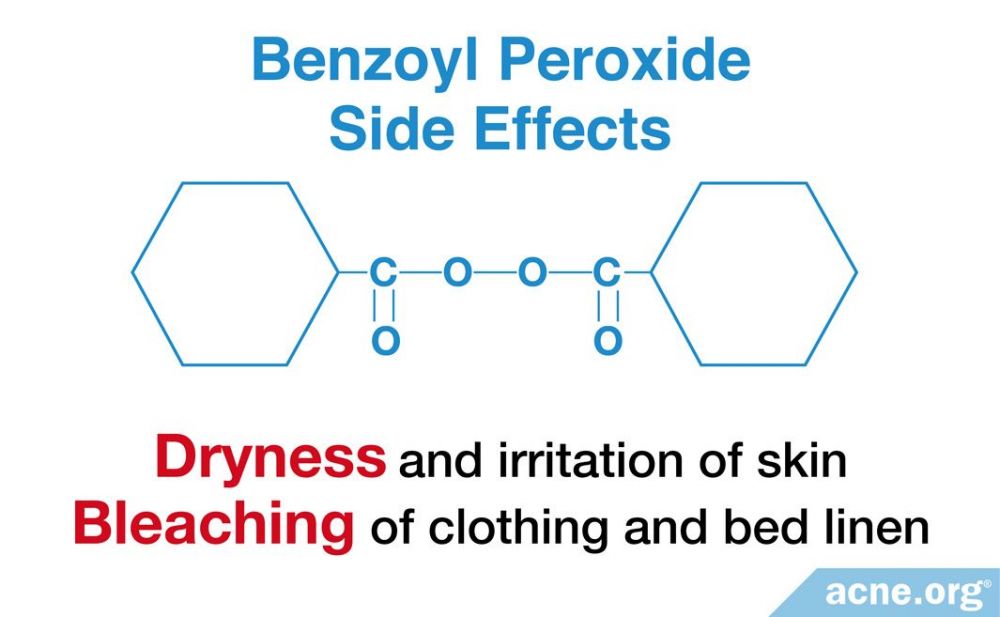
Benzoyl peroxide has two common side effects that may cause problems for patients:
- Dryness and irritation of skin
- Bleaching of clothing and bed linen that benzoyl peroxide may contact
Patients may stop treatment after a few days if they experience these effects. However, when patients are aware of the potential side effects and get advice on how to minimize these effects they usually stay with the treatment long enough to see their acne clear up.1
One strategy is to use a lower strength of benzoyl peroxide. 10% benzoyl peroxide has greater side effects than 2.5% benzoyl peroxide, but the 2.5% strength reduces acne just as effectively. Another advantage of 2.5% benzoyl peroxide is that the side effects decrease as treatment continues.3,10
Recent research also shows that applying vitamin E before using benzoyl peroxide decreases side effects while improving the treatment of acne, but the best method for achieving this remains to be seen.11
Patients can use this information and develop best practices to manage the side effects. Most people find that starting with a small amount of benzoyl peroxide and using it every other day is a good way to start, and researchers agree.10
Benzoyl peroxide is available in gels, aquagels, creams, lotions, and washes. Washes usually do not clear the skin as much as leave-on products like gels, creams, and lotions.
Regarding bleaching of fabrics, benzoyl peroxide should be allowed to dry before coming in contact with clothing or bed linens to prevent bleaching.3
Allergic contact dermatitis is a potentially more serious side effect of benzoyl peroxide. It is rare, occurring in only one out of every five hundred patients, but if you see swelling and/or severe crusting of the skin, this could be a sign of allergy, in which case you should immediately cease using benzoyl peroxide.3
References
- Tucker, R. & Walton, S. The role of benzoyl peroxide in the new treatment paradigm for acne. Pharm. J. 279, 48 – 53 (2007). https://www.ncbi.nlm.nih.gov/pubmed/23839205
- Mohammad, T. F. Acne therapeutics: A closer look at Benzoyl Peroxide. Skinmed 13, 94 – 96 (2015). https://www.ncbi.nlm.nih.gov/pubmed/26137733
- Sagransky, M., Yentzer, B. A. & Feldman, S. R. Benzoyl peroxide: a review of its current use in the treatment of acne vulgaris. Expert Opin. Pharmacother. 10, 2555 – 2562 (2009). https://www.ncbi.nlm.nih.gov/pubmed/19761357
- Thiele, J. J. Oxidative targets in the stratum corneum. A new basis for antioxidative strategies. Skin Pharmacol. Appl. Skin Physiol. 14 Suppl 1, 87 – 91 (2001). https://www.ncbi.nlm.nih.gov/pubmed/11509912
- Bellei, E., Rota, C., Bergamini, S., Manfredini, P., Albertazzi, A., Tomasi, A. & Iannone, A. Effect of alpha-tocopherol and N-acetylcysteine on benzoyl peroxide toxicity in human keratinocytes. J. Biochem. Mol. Toxicol. 18, 107-114 (2004). https://pubmed.ncbi.nlm.nih.gov/15122653/
- Okamoto, K., Kanayama, S., Ikeda, F., Fujikawa, K., Fujiwara, S., Nozawa, N., Mori, S., Matsumoto, T., Hayashi, N. & Oda, M. Broad spectrum in vitro microbicidal activity of benzoyl peroxide against microorganisms related to cutaneous diseases. J. Dermatol. 48, 551-555 (2021). https://pubmed.ncbi.nlm.nih.gov/33369759/
- Stinco, G., Bragadin, G., Trotter, D., Pillon, B. & Patrone, P. Relationship between sebostatic activity, tolerability and efficacy of three topical drugs to treat mild to moderate acne. J. Eur. Acad. Dermatology Venereol. 21, 320 – 325 (2007). https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1468-3083.2006.01914.x
- Cunliffe, W. J., Stainton, C. & Forster, R. A. Topical benzoyl peroxide increases the sebum excretion rate in patients with acne. Br. J. Dermatol. 109, 577 – 579 (1983). https://www.ncbi.nlm.nih.gov/pubmed/6227330
- Zuliani, T., Khammari, A., Chaussy, H., Knol, A. C. & Dréno, B. Ex vivo demonstration of a synergistic effect of adapalene and benzoyl peroxide on inflammatory acne lesions. Exp. Dermatol. 20, 850 – 853 (2011). https://www.ncbi.nlm.nih.gov/pubmed/21793939
- Tripathi, S. V, Gustafson, C. J., Huang, K. E. & Feldman, S. R. Side effects of common acne treatments. Expert Opin. Drug Saf. 12, 39 – 51 (2013). https://www.ncbi.nlm.nih.gov/pubmed/23163336
- Mills, O. H., Criscito, M. C., Schlesinger, T. E., Verdicchio, R. & Szoke, E. Addressing free radical oxidation in acne vulgaris. J. Clin. Aesthet. Dermatol. 9, 25 – 30 (2016). https://www.ncbi.nlm.nih.gov/pubmed/26962389
 Acne.org Products
Acne.org Products