It Is Most Likely Best to Avoid Using Baking Soda on Acne-Prone Skin

The Essential Info
Reasons to Use Baking Soda for Acne
- Baking soda may have some weak antibacterial properties.
Reasons Not to Use Baking Soda for Acne
- When baking soda is applied to the skin, it can be abrasive. This could damage the skin and cause irritation. It is well known that physically irritating the skin can lead to more breakouts.
- Baking soda has an alkaline pH that does not match the skin’s naturally acidic pH. For acne prevention, the skin’s acidic pH should be maintained. Therefore, baking soda could be detrimental to acne-prone skin by raising the pH of the skin.
The Bottom Line: When we take the evidence as a whole, it is most likely best to avoid using baking soda on acne-prone skin.

The Science
Baking soda, also known as sodium bicarbonate (NaHCO3), is a common household product used in baked goods, toothpaste, for cleaning, odor control, and sometimes even as an antacid. Many people also wonder if it might help with acne by mixing it with water and applying it as a paste on acne-affected areas.1
Scientists have never performed a study directly on whether baking soda might help with acne. The only clinical evidence we have regarding baking soda and skin disease comes from one study where researchers looked at whether topical application of sodium bicarbonate would help with symptoms of psoriasis, which is a different skin disease from acne, but has some commonalities such as redness and impaired skin function. The study found that applying sodium bicarbonate to the skin had no beneficial effect on psoriasis.2
Since we only have that small bit of underwhelming evidence from clinical studies, let’s dive deeper and have a look at studies investigating the properties of baking soda and try to infer how these properties may impact acne. As we will see, while baking soda may have a weak antibacterial effect, as a whole, the evidence shows us that acne-prone people should most likely avoid using it on their skin.3-4
The three most commonly studied properties of baking soda are:
- Antibacterial effect – very slight, unlikely to provide much benefit
- Exfoliating properties – could cause physical irritation of the skin
- Alkaline nature – could change the natural acidity of the skin
Let’s have a look at these one by one.
Antibacterial Effect
Acne is a disease that is made worse by the bacteria C. acnes. There are no studies to evaluate whether baking soda has antibacterial effects on C. acnes.
However, a 2005 study published in the Journal of Dental Hygiene evaluating baking soda in toothpaste demonstrated that baking soda has weak antibacterial properties against the bacterium S. mutans, which is a bacterium of similar class and characteristics as C. acnes. In the study, baking soda was not able to kill S. mutans, but showed potential in limiting its growth.
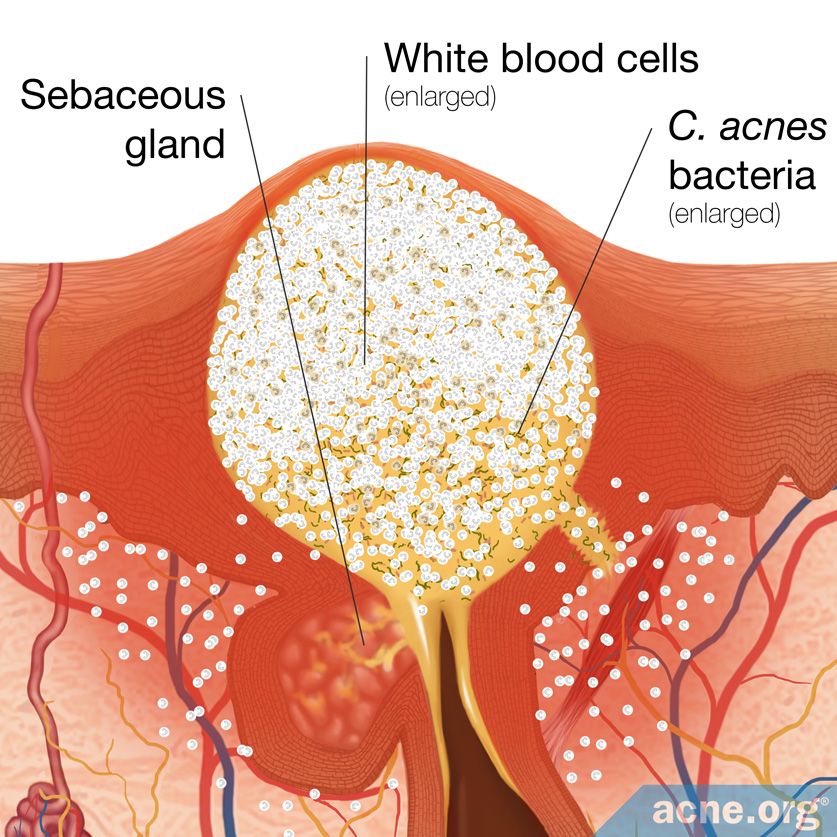
When compared with many other compounds that can kill bacteria, simply limiting the growth of bacteria is unimpressive. So even though baking soda might limit the growth of C. acnes bacteria, this is cause for limited hope.
Click here for details on the study

To perform this study, the researchers grew S. mutans in a laboratory and exposed the bacteria to either baking soda or hydrogen peroxide. The study found that baking soda prevented S. mutans growth, while hydrogen peroxide killed S. mutans. Therefore, the scientists concluded that baking soda has the ability to inhibit bacterial growth.
Most individuals with acne have an overgrowth of Cutibacterium acnes (C. acnes) bacteria in their skin. Although not directly studied in this experiment, it is possible that baking soda may also inhibit the growth of C. acnes bacteria. So, in a best-case scenario, would baking soda just halt the growth of C. acnes, and if so, would that help lessen acne symptoms? Perhaps to a limited degree. However, scientists will need to perform more studies to confirm this hypothesis.4
Exfoliating Properties
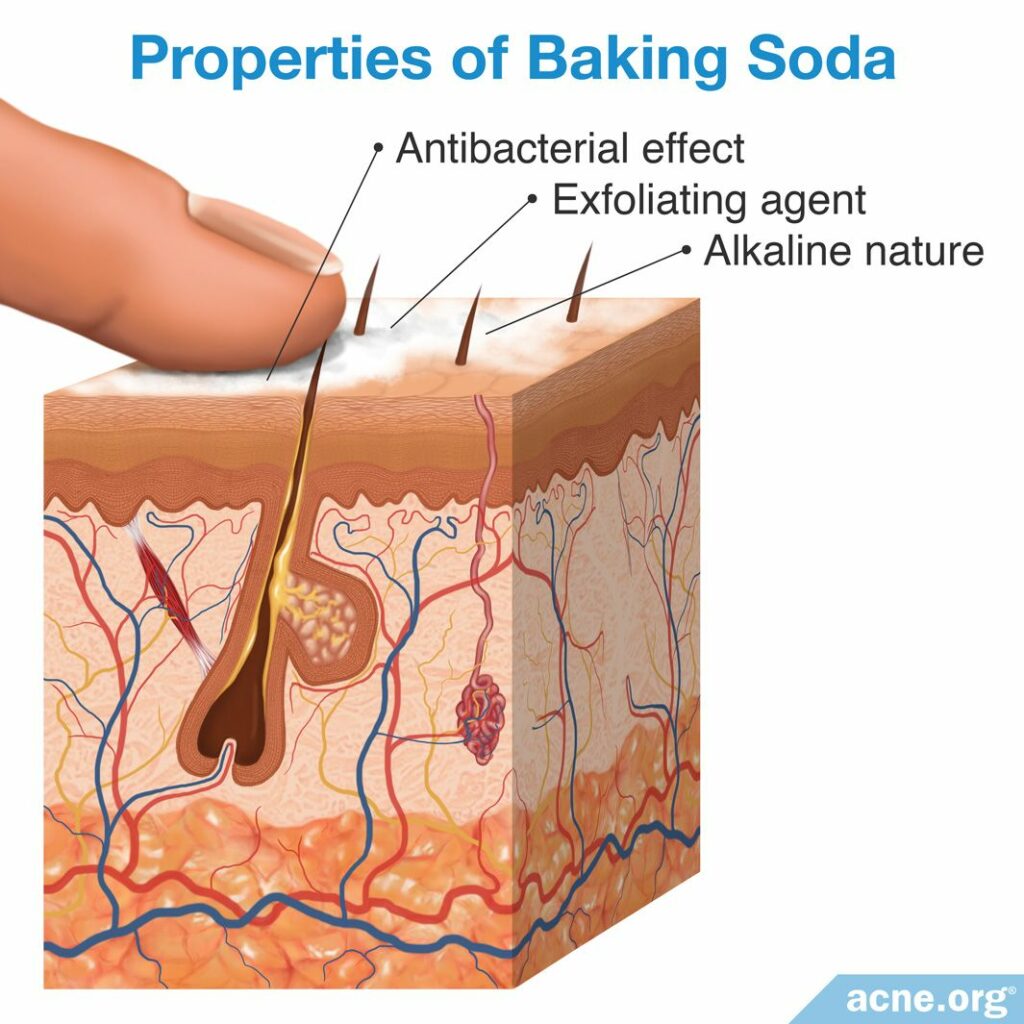
Baking soda acts as an abrasive, which means that it is able to remove substances that are stuck on a surface. For instance, in toothpaste, the abrasive nature of baking soda is used to remove plaque buildup on teeth.
When used on the skin, the abrasive quality of baking soda acts as an exfoliating agent to remove dead skin cells. Acne is formed through the accumulation of substances, including dead skin cells, in the pores. Theoretically, if you exfoliate the surface of the skin in a non-irritating way, this can signal the skin underneath to produce cells more quickly, which can help prevent clogs.4,5
However, the key is to exfoliate in a non-irritating way. When it comes to baking soda, its abrasive quality means it is potentially irritating to the skin. It is well-known that anything that irritates the skin can cause more acne. Therefore, exfoliating the skin with baking soda could cause irritation and potentially make breakouts worse. Instead of physically exfoliating with an abrasive like baking soda, it is far more effective to exfoliate chemically with an acid, like glycolic acid, which is non-irritating and has been proven to help with acne.
Alkaline Nature
Baking soda has a high pH (~9.0), which means it is alkaline or “basic” in nature. The skin, on the other hand, is naturally acidic (~5.5).
This means that when used on the skin, baking soda may cause the skin to become more alkaline. Some skin care bloggers suggest that using alkaline skin products will make the skin healthier, but this is not true. Changing the skin’s pH may impair the skin’s barrier.6 For ideal skin health, and prevention of acne, the skin’s barrier should be left as intact as possible. And therefore, the pH should be left unchanged.
In short, since using baking soda on the skin can raise its pH, this is very likely a bad idea when it comes to acne.
Bottom Line
The weak antibacterial activity of baking soda is unlikely to significantly improve acne, and its abrasive nature and alkaline pH may in fact lead to more breakouts, not less.
References
- Yoon, S.-K. A Friend In need. Far East. Econ. Rev. 165, 60 (2002). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2806659/
- Mazzarello, V., Piu, G., Ferrari, M. & Piga, G. Efficacy of a Topical Formulation of Sodium Bicarbonate in Mild to Moderate Stable Plaque Psoriasis: a Randomized, Blinded, Intrapatient, Controlled Study. Dermatol Ther (Heidelb). 9, 497-503 (2019). https://www.ncbi.nlm.nih.gov/pubmed/31077089
- Al-Abri, S. A. & Kearney, T. Baking soda misuse as a home remedy: case experience of the California Poison Control System. J. Clin. Pharm. Ther. 39, 73 – 77 (2014). https://www.ncbi.nlm.nih.gov/pubmed/24313600
- Silhacek, K. J. & Taake, K. R. Sodium bicarbonate and hydrogen peroxide: the effect on the growth of Streptococcus mutans. J. Dent. Hyg. 79, (2005). https://www.ncbi.nlm.nih.gov/pubmed/16297309
- Thong, S., Hooper, W., Xu, Y., Ghassemi, A. & Winston, A. Enhancement of plaque removal by baking soda toothpastes from less accessible areas in the dentition. J. Clin. Dent. 22, 171-178 (2011). https://www.ncbi.nlm.nih.gov/pubmed/22403983
- Hachem, J., et al. pH directly regulates epidermal permeability barrier homeostasis, and stratum corneum integrity/cohesion. The Journal of Investigative Dermatology 121, 345-353 (2003). https://pubmed.ncbi.nlm.nih.gov/12880427/
 Acne.org Products
Acne.org Products